Segregation of Impurity Atoms
Images of their surface structures
Segregation is the enrichment of an atomic species in one phase of a multiphase system at the surface or at an interface (e.g. a grain boundary between different crystallites in a metal). By STM, we can study surface segregation, i.e., the enrichment at the surface of atoms which are present in the bulk material (but in smaller concentrations there). Some impurities, such as carbon, sulfur and phosphorus, can reach concentrations in the percent range at the surface or an interface even though their bulk concentration is only a few parts per million (ppm).
In many cases, segregation of atoms like oxygen or nitrogen from the bulk is simply an alternative to adsorption from the gas phase, sometimes it cannot be avoided because no one can manufacture a crystal pure enough to avoid impurity segregation. Impurity segregation to so-called inner surfaces also has immense importance in technology as mentioned below.
Carbon - sometimes white, coloring other atoms dark
On metal surfaces, carbon impurity atoms usually occupy sites between the metal atoms. Being much smaller than metal atoms, the carbon atoms are usually invisible to STM. Only under special tip conditions, they become visible as bright spots as in this image of segregated C on a PtNi alloy surface:

Carbon atoms are circled in part of this image. The single bright rows (shifted rows of metal atoms) are due a surface reconstruction unrelated to the segregated carbon. In this image it is not possible to distinguish between Pt and Ni atoms, but there is a clear difference between metal atoms without carbon neighbours (brightest), those with one carbon neighbour (grey) or two carbon neighbours (darkest): In the region marked 'p(2×2)' in the image, squares of 2×2 metal atoms have C atoms at the corner (one C neighbour), whereas there are twice as much C atoms in the darker c(2×2) areas. This reduction of apparent height by C atoms is not a geometric effect but due to a reduced electron density reducing the tunneling current in the STM.
Usually, C atoms are not imaged, but only their influence on the apparent height of metal atoms can be used to detect them:

So it looks as if carbon (sitting between the metal atoms) colors the adjacent metal atoms dark in our STM images.
What should be in transformer steel and what shouldn't
Segregation of impurities to grain boundaries in steel can cause brittleness and other undesirable phenomena. With STM, we cannot look at grain boundaries (“inner surfaces”) but we can study the (outer) surface to find out processes and effects which may work at inner surfaces as well.
The following image shows the (110) surface of Fe-3.5at% (“transformer steel”). Nearly 1/3 of all surface atoms are silicon (dark) replacing iron atoms. Furthermore, carbon betrays its presence by making the iron atoms in the central rows of the ladder-like structures invisible (see structure model for the rectangular region).
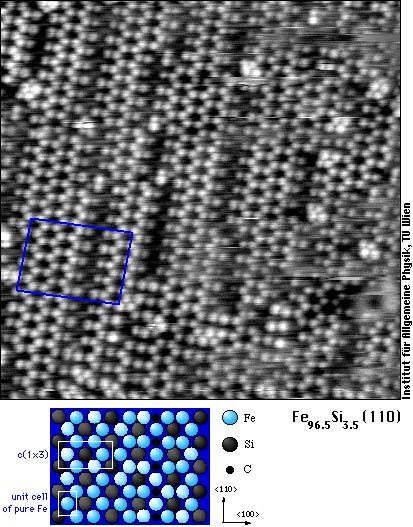
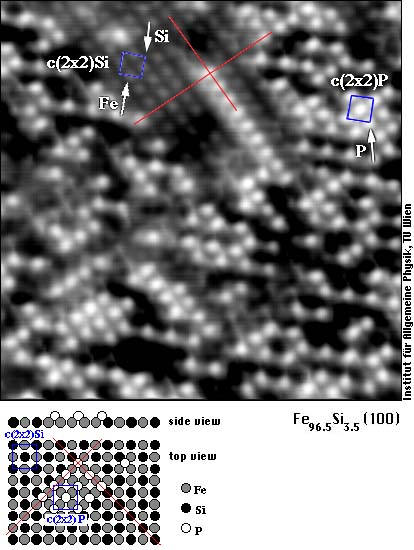
Silicon is desired in transformer steel, alloying it into the material greatly reduces the electrical losses in transformers and motors. Phosphorous is one of those unwanted impurities that segregate to grain boundaries and surfaces, even when present in only minute concentrations in the bulk. We find that it also sits in a different position. Whereas Si replaces iron atoms and C sits between them, phosphorus atoms are situated above the surface and are imaged as white dots. You can clearly see this on the following image of a (100) oriented surface of the same alloy.
For further information
- C on PtNi
M. Schmid, A. Biedermann and P. Varga
Segregated carbon on Pt10Ni90(100) studied by scanning tunneling microscopy
Surf. Sci. 294 (1993) L952-L958. Full text*
- Si and on Fe(110)
A. Biedermann, M. Schmid and P. Varga
Segregated Si on Fe96.5Si3.5(110): domain wall structures in a two-dimensional alloy
Phys. Rev. B 50 (1994) 17518-17524. Full text*
- Si and C on Fe(110)
A. Biedermann, M. Schmid and P. Varga
Domain wall structures in an ordered Si/Fe(110) surface alloy
Surf. Sci. 331-333 (1995) 787-793.
- Si and P on Fe(100) and Fe(110)
A. Biedermann, M. Schmid, B. M. Reichl and P. Varga
Competitive segregation of Si and P on Fe96.5Si3.5(100) and (110)
Fresenius J. Anal. Chem. 353 (1995) 259-262.
* Please note: access to full text (PDF files) of some articles is restricted due to copyright reasons.
This page, including all the errors and typos, was written by:
Michael Schmid, IAP/TU Wien ([email address: lastname @ this server · enable javascript to see it]).


